Roche files vemurafenib for metastatic melanoma in Europe and US
Yesterday, Roche/Genentech announced that they have submitted the New Drug Application (NDA) filing for PLX4032 (vemurafenib) in BRAF V600E mutation-positive metastatic melanoma, based on BRIM2 and BRIM3 trials to both the FDA and EMA.
I’ve posted quite a bit on BRAF inhibitors such as vemurafenib on this blog – check out the related posts feature at the bottom if you want to learn more about the history of this class of drugs.
Interestingly, Roche have also submitted the companion diagnostic, cobas 4800 BRAF V600 Mutation Test, which means that oncologists in the community will be able to more easily test patients for the mutation, since these patients are more likely to respond to therapy with BRAF inhibitors such as vemurafenib.
Although melanoma is treatable in the early stages, unfortunately once it becomes metastatic, it becomes more difficult to contain.
Roche have conducted two large-scale clinical trials (BRIM3 and BRIM2) to evaluate the safety and effectiveness of vemurafenib in mestatatic melanoma patients who have the V600E mutation. According to the Company:
“BRIM3 is a global, randomised, open-label, controlled, multicentre, Phase III study that compared vemurafenib to dacarbazine chemotherapy, a current standard of care, in 675 patients with previously untreated BRAF V600 mutation-positive, unresected or locally advanced metastatic melanoma.
The study met its two co-primary endpoints and showed that participants who received vemurafenib lived longer (overall survival) and also lived longer without their disease getting worse (progression-free survival) compared to those who received dacarbazine chemotherapy.
The safety profile was consistent with previous vemurafenib studies.”
The BRIM2 study also formed part of the global filing:
“BRIM2 is a global, single-arm, multicentre, open-label Phase II study that enrolled 132 patients with previously treated BRAF V600 mutation-positive metastatic melanoma. The primary endpoint of the study was overall response rate as assessed by an independent review committee.
The study showed that vemurafenib shrank tumours in 52 percent of trial participants. People who participated in the trial lived a median of 6.2 months without their disease getting worse (median PFS).”
Like many of us, I’ve known family and friends or friends of friends who have been lost to this devastating disease. Six months doesn’t sound very long, but when you look at many of the oncologic filings over the last decade, very few have actually shown six months improvement in overall survival. In that context, this represents a dramatic advance for patients.
Of course, we can still do better, and as we learn more about the biology of the disease, so we can develop smarter therapeutic strategies for overcoming malignant melanoma. This could take the form of new combinations or other BRAF inhibitors that do not target CRAF as well as BRAF, for example:
“The most frequent Grade 3 adverse event observed in clinical trials of vemurafenib was cutaneous squamous cell carcinoma, a common skin cancer treated by local excision (minor surgery done in a physician’s office).
The most common adverse events were rash, photosensitivity, joint pain, hair loss and fatigue.”
It has been shown that activating CRAF, for example, is most likely responsible for the squamous cell proliferation on the skin in some patients. It appears to be reversible once treatment is stopped. However, that said, this is still a huge advance and should be applauded as such.
More BRAF and RAF inhibitors are on the way
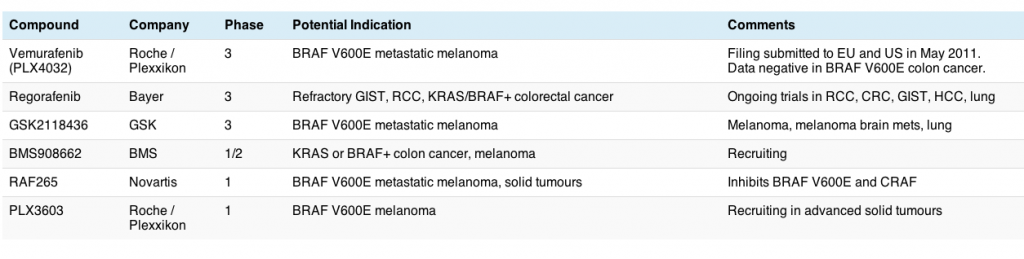
Interestingly, Bayer also announced yesterday that their multi-kinase RAF/VEGF/Tie2/RET/KIT inhibitor, regorafenib, was granted Fast Track designation by the FDA for the treatment of patients with metastatic and/or unresectable gastrointestinal stromal tumors (GIST) whose disease has progressed despite at least imatinib and sunitinib as prior treatments. This is an incredibly small population so I don’t expect the trial to accrue speedily. GIST is largely KIT or PDGF rather than RAF driven. The trial began enrolling in January this year and accrual is still open – in all, Bayer plan to enroll 170 patients.
I will be interested to see whether regorafenib also activates CRAF as well as RAF – if so, we can expect some squamous cell proliferation in the adverse events – while multi-kinase inhibitors can be effective, we also have seen some off-kinase side effects, as shown with sorafenib and sunitinib.
What does this data mean?
The vemurafenib filing is excellent news for patients based on the data and pictures I have seen of responses in various conference presentations to date. It’s a great success story of finding an aberrant mutation, developing a diagnostic to enable widespread testing for it and matching the patient to the treatment.
What I also like is the fervour with which the melanoma scientist-physician community is researching the methods of resistance and looking at new combinations with BRAF inhibitors almost as soon as they detect them – a great showcase for what to do.
We can also expect update on the BRIM2 and 3 trials at the forthcoming annual meeting at ASCO next month – I can’t wait to see how the data is maturing!
Assuming that vemurafenib is approved, it will also be interesting to see what happens post-approval, since Daiichi Sankyo bought Plexikkon, complicating the potential marketing and sales roll-out considerably. One can wonder at the old adage that two’s company, but three becomes a crowd to manage.
7 Responses to “Roche files vemurafenib for metastatic melanoma in Europe and US”
Thanks for this post, Sally. The chart is really helpful. – Elaine
Thanks, Elaine – it’s a nifty tool that saves a lot of blah 🙂
Sally, also remember Roche is likely to be filing an NDA for GDC-0449 in basal cell carcinoma in both the US and ED this year. Sounds like they have two great drugs for skin cancer on the slate.
Yes indeed, Craig. I saw Ervin Epstein’s BCC presentation live at the recent AACR meeting – stunning data. I’m certainly not going to forget the photographs of the responses in some of his patients in a long while. I didn’t mention it because this post is really about metastatic melanoma and BRAF inhibition, specifically. However, you are correct, we will hopefully see two filings in very different types of skin cancer from Roche this year.
[…] Ipilimumab (considered here), now sold as Yervoy. Just yesterday, as considered in the Pharma Strategy blog (with a helpful chart of BRAF inhibitors), Roche/Genentech submitted an application to the FDA […]
This was really helpful. But, how many of these trials were tested on 84 year olds, with matastesized brain cancer? Originating from Melanoma of on their lymph nodes? Please comment so we can decide whether to proceed with this type of treatment….thanks
Cindy, I have no idea without seeing the data. Typically, clinical trials are done in younger patients although it all depends upon performance status of patient. I would chat with your oncologist about the options available to you.
Comments are closed.