Aside from the already published Part 1 and Part 2 blog posts about AACR here on PSB, you can also find some more coverage, including summaries of other topics, on the companion Biotech Strategy Blog, such as the following:
A new development in R&D – hypoxia-activated prodrug (HAP)?
Today I wanted to discuss a completely different subject though. Some data that was eagerly awaited prior to the meeting was the Threshold Pharma data for TH–302 in pancreatic cancer. The concept is an interesting one theoretically – simply described, they sought to develop a compound that is specifically activated under hypoxic conditions – and look for more potent anti-cancer activity.
Now, preclinically, the data looked impressive – see Sun et al., (2011) for a comprehensive overview (reference below, open access) and certainly bore out the hypothesis from a conceptual standpoint, but does this translate into the clinic with patients?
It should be noted that most normal cells in the body thrive under normoxic conditions, whereas tumours tend to grow through angiogenesis under more extreme conditions, with patches of hypoxia and normoxia.
One of the challenges with most existing chemotherapies is getting enough of the drug inside the tumour to cause enough of an effect. Because they are non-specific, they often attack a lot of normal cells in their path too.
Threshold’s theory is essentially based on a pro-drug approach; such bioreductive agents are activated under hypoxic conditions and are not a new concept in cancer research – mitomycin C is a classic one of this genre, for example. They state that:
“After conversion to the active form of the drug, the more resistant hypoxic cells are exposed to high concentrations of released cytotoxic agent, which can also diffuse into the surrounding oxygenated regions of the tumor, exerting what is referred to as a bystander effect.
In this way, TH–302 can kill more of the tumor than can otherwise be accounted for by the hypoxic fraction alone. Because of its selective activation in the hypoxic regions of solid tumors, we believe that TH–302 will be less likely to produce the systemic toxicity caused by most cytotoxic chemotherapies.”
Chemotherapy, however, can also induce a more hypoxic environment and paradoxically induce the production of more cancer stem cells (CSCs), as shown in this excellent graphic published in The Scientist recently by Liu et al., (2012):

Source: The Scientist
As Liu et al., (2012) observed:
“These treatments can create inflammation in the tissue surrounding the tumor as well as hypoxia, or loss of oxygen, which activates Wnt signaling. Inflammatory mediators such as IL–8, IL–6, and Wnt signaling spur CSCs to self-renew or increase in number, thus driving tumor growth.”
Thus while trying to shrink the tumour, there may be some cases where hypoxia induced by chemotherapy can make the tumour more aggressive. This is one of conundrums of cancer chemotherapy. For those of you interested in reading more on CSCs, check out Joan Massaugué’s impressive research for more information.
Ok so far, but what happens in practice?
In this phase IIb trial, patients with advanced pancreatic cancer (n=214), including 77% with distant metastases, were randomised to receive either the standard of care, gemcitabine, TH–302 (240 mg/m2) plus gemcitabine, or TH–302 (340 mg/m2) plus gemcitabine.
Let me state upfront – there were no overall survival (OS) available in the pancreatic study, so we only have progression-free survival (PFS) to go on for now. Surrogate endpoints such as PFS do not always equate to OS, ie patients living longer. With that caveat, the main data can be summarised as thus:
- PFS increased from 3.6 months (gemcitabine alone) to 5.6 months (gem + TH–302, either dose).
- The hazard ratio comparing the TH–302 combination to gemcitabine alone was 0.61 (p = 0.005).
- There was also an increase in tumor response rate from 12% to 22%.
- TH–302 related toxicities included myelosuppression, skin and mucosal toxicities, which were dose dependent and consistent with previous trials.
While the study achieved it’s primary endpoint, it should be noted that there are some issues with extrapolating limited phase IIb data to potential success in a phase III trial. For starters, you want to have your groups as carefully balanced as possible or you could be making a leap of faith based on the wrong premise. Malcolm Moore alluded to this fact in his discussion of the data. Looking at the baseline characteristics, I was dismayed to see several areas where the groups weren’t well balanced, which is sloppy research – don’t give people a reason to doubt the data before you start:
- Performance status – the TH–302 arms had slightly better PS
- The percentage of patients over 65 in the gm +TH–302 240mg/m2 was noticeably less than the other two arms (39% vs 59% and 51%)
- The gem alone arm had a higher number of patients with liver mets (67% vs 63 and 57%), which could be argued as more advanced and difficult to treat
- Baseline CA19–9 was higher in both TH–302 arms (doubled, why?)
- Lower dose arm of TH–302 had less prior systemic treatment in the adjuvant or neoadjuvant setting (8% vs 13% and 11%)
- Treatment cycles – the group receiving TH–302 had a median of 2 more cycles and thus longer follow up
Could these factors influence the results? Unfortunately yes, especially when several were weighted in favour of the TH–302 treatment groups. Should they have made a better attempt at balancing them? Absolutely – it’s been a while since I saw so many imbalances and these go both ways making it hard to extrapolate anything much from the efficacy data – it detracts from the study. I wouldn’t like to guess how these might influence a larger, randomised trial in more centres where any efficacy signal seen in phase IIb can rapidly disappear when such bias is removed or significantly reduced.
The bigger challenge, though, as Malcolm Moore pointed out is that the absolute PFS benefit was only 2 months with TH–302 therapy (240mg/m2 dose), which is probably on the limit for moving forwards to a phase III randomised trial.
Overall
I think this is a nice scientific idea that is well worth testing, but TH–302 may not be the one that gets the breakthrough status based on these limited results. Future compounds may be able to induce more potency and efficacy in difficult to treat tumour types – a 2 month absolute benefit in a phase II trial can rapidly disappear in a large scale randomised study, which is something I would be concerned about given some of the imbalances in the patient groups to begin with.
A phase III SPA has been agreed with the FDA in soft-tissue sarcomas (STS), but regular readers will be well aware that I’m not a fan of catch-all studies in unselected patients, especially in broad heteregeneous cancers such as sarcomas and pancreatic cancer. That’s just asking for trouble because the non-responders will lower the overall efficacy, as we saw recently with ridaforolimus, and this sets up a trial for almost certain failure unless smaller subsets with more clearly defined targets can be identified.
References:
 Sun, J., Liu, Q., Wang, J., Ahluwalia, D., Ferraro, D., Wang, Y., Duan, J., Ammons, W., Curd, J., Matteucci, M., & Hart, C. (2011). Selective Tumor Hypoxia Targeting by Hypoxia-Activated Prodrug TH–302 Inhibits Tumor Growth in Preclinical Models of Cancer Clinical Cancer Research, 18 (3), 758–770 DOI: 10.1158/1078–0432.CCR–11–1980
Sun, J., Liu, Q., Wang, J., Ahluwalia, D., Ferraro, D., Wang, Y., Duan, J., Ammons, W., Curd, J., Matteucci, M., & Hart, C. (2011). Selective Tumor Hypoxia Targeting by Hypoxia-Activated Prodrug TH–302 Inhibits Tumor Growth in Preclinical Models of Cancer Clinical Cancer Research, 18 (3), 758–770 DOI: 10.1158/1078–0432.CCR–11–1980
 At first view, I was impressed by the enormous organization and very large number of participants, which was at least as important as the ASCO annual meeting. However, as a clinician, I only recognized very few familiar faces as the large majority of attendance included basic and translational scientists, as well as representatives of pharma.
At first view, I was impressed by the enormous organization and very large number of participants, which was at least as important as the ASCO annual meeting. However, as a clinician, I only recognized very few familiar faces as the large majority of attendance included basic and translational scientists, as well as representatives of pharma.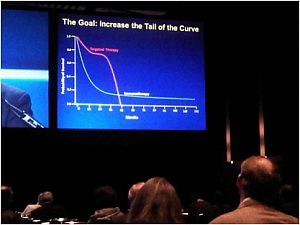 The goal is to increase the tail of the curve in the photograph in the right. The approval of ipilimumab in the treatment of metastatic melanoma has inaugurated the new era of anti-cancer immune therapies.
The goal is to increase the tail of the curve in the photograph in the right. The approval of ipilimumab in the treatment of metastatic melanoma has inaugurated the new era of anti-cancer immune therapies.