Some additional thoughts on Zometa in early breast cancer
Yesterday’s news from the AZURE trial at the San Antonio Breast Cancer Symposium generated a lot of buzz. I posted the data after the press briefing in the afternoon here but have received a lot of questions from people confused by the press coverage of the data. To be fair, the physician presenting the data, Dr Robert Coleman, didn’t help matters by stating:
“Clearly our results are very different from those published by the ABCSG investigators”
when referring to the previous study published in the NEJM in February last year, without elaborating clearly on the differences. Dr Coleman also stated that it was “a negative trial”.
Interestingly, in the panel disclosures only Dr Giordano from MD Anderson revealed nothing to declare; Drs Coleman (Novartis and Amgen), Chlebowski (Novartis and Amgen) and Gnant (Novartis, Amgen and AstraZeneca) all received industry funding of some kind.
For those of you interested in the detail, I included the journal reference and the differences between the two trials in the post yesterday.
But to simplify – they are actually two very different trials – involving different stages of disease, menopausal status and different base treatments, so in reality, we should not expect them to be the same, since the populations are different.
Here’s a quick summary in the table below:
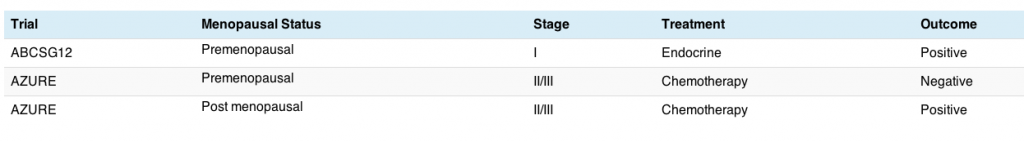
What we can see from this is that the stage II/III premenopausal women who received chemotherapy did not do well on zoledronic acid, but the other two groups did. I should point out that the post menopausal subset group who did benefit were typically more than 5 years post menopause and amounted to around one third of the study participants, so it was not an insignificant number. Lumping both menopausal ER+ subgroups together for the purpose of an overall analysis is fraught with challenges given the potential heterogeneity if the two behave differently, thereby effectively cancelling each other out.
We can also see that comparing the premenopausal groups in the two trials is not an easy comparison since one received hormonal therapy and the other chemotherapy. Taking hormonal therapy essentially induces early menopause, so this means that the ABCSG trial participants may actually model the AZURE postmenopausal women closer than people realise.
The whys and wherefores will not doubt follow in due course from the breast cancer oncologists.
My only surprise in all this is that Novartis have already stated they are withdrawing the filing, which may be a little hasty given that they have 2 out of 3 positive subsets of women with breast cancer who appear to significantly benefit from zoledronic acid treatment.
Ah well, time will tell where this controversy will lead!
2 Responses to “Some additional thoughts on Zometa in early breast cancer”
Sally,
It’s been really important to me to follow you through the ins and outs of this trial — or these two very different trials. Obviously there were comparisons made when the two were not comparable and the issues for the many women surviving breast cancer are complex. From the beginning I found the findings not just confusing but confounding given:
1) some 70% of recurrences in early BC occur in the bone;
2) estrogen-reducing therapies accelerate bone loss (both chemo and aromastase inhibitors)
3) the “transition” period in younger, pre-menopausal women who are given both chemo and estrogen-blocking drugs can cause a severe and sudden drop in bone density — wouldn’t that, in and of itself – create an environment more conducive to metastatic activity?
I don’t know the answers, because I’m not a scientist. That is why I’m grateful for the opportunity to pester you with questions. There is a lot at stake for many in this trial and I’m certainly hope we haven’t heard that last of it when the conference ends this weekend. We all deserve better. Thanks for taking the effort —
Jody
Thanks for the link to the article.I did want to know a lot about breast cancer as a close family member is stricken and I want to extend as much help as possible other than love and kindness.
Comments are closed.